Calcium-induced calcium release during action potential firing in developing inner hair cell
Authors
Iosub, R., Avitabile, D., Grant, L., Tsaneva-Atanasova, K. and Kennedy, H.J.
Abstract
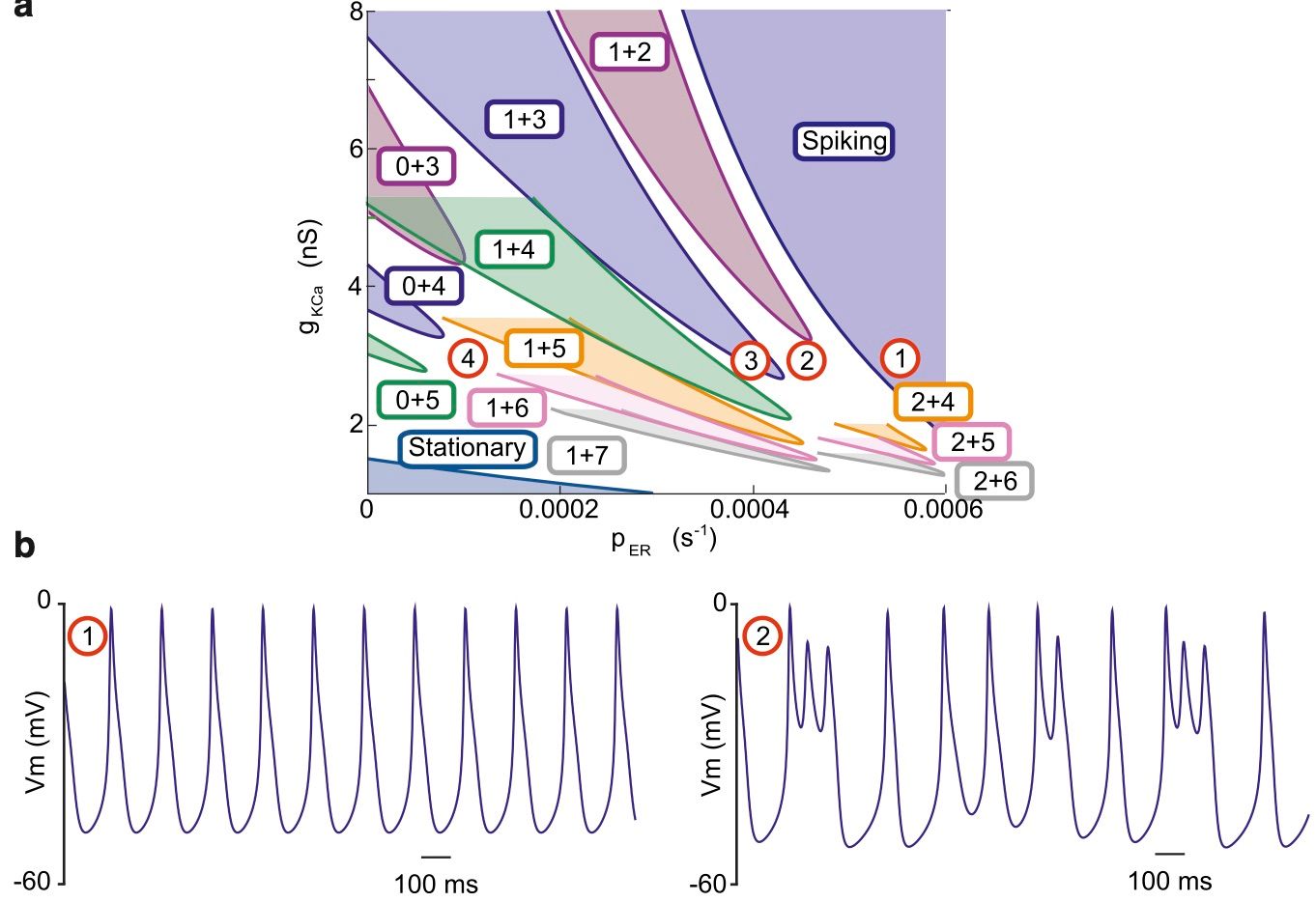
In the mature auditory system, inner hair cells (IHCs) convert sound-induced vibrations into electrical signals that are relayed to the central nervous system via auditory afferents. Before the cochlea can respond to normal sound levels, developing IHCs fire calcium-based action potentials that disappear close to the onset of hearing. Action potential firing triggers transmitter release from the immature IHC that in turn generates experience-independent firing in auditory neurons. These early signaling events are thought to be essential for the organization and development of the auditory system and hair cells. A critical component of the action potential is the rise in intracellular calcium that activates both small conductance potassium channels essential during membrane repolarization, and triggers transmitter release from the cell. Whether this calcium signal is generated by calcium influx or requires calcium-induced calcium release (CICR) is not yet known. IHCs can generate CICR, but to date its physiological role has remained unclear. Here, we used high and low concentrations of ryanodine to block or enhance CICR to determine whether calcium release from intracellular stores affected action potential waveform, interspike interval, or changes in membrane capacitance during development of mouse IHCs. Blocking CICR resulted in mixed action potential waveforms with both brief and prolonged oscillations in membrane potential and intracellular calcium. This mixed behavior is captured well by our mathematical model of IHC electrical activity. We perform two-parameter bifurcation analysis of the model that predicts the dependence of IHCs firing patterns on the level of activation of two parameters, the SK2 channels activation and CICR rate. Our data show that CICR forms an important component of the calcium signal that shapes action potentials and regulates firing patterns, but is not involved directly in triggering exocytosis. These data provide important insights into the calcium signaling mechanisms involved in early developmental processes.
Links
DOI PDFBibTeX
@article{iosub2015calcium,
title={Calcium-induced calcium release during action potential firing in developing inner hair cells},
author={Iosub, Radu and Avitabile, Daniele and Grant, Lisa and Tsaneva-Atanasova, Krasimira and Kennedy, Helen J},
journal={Biophysical journal},
volume={108},
number={5},
pages={1003--1012},
year={2015},
publisher={Elsevier}
}